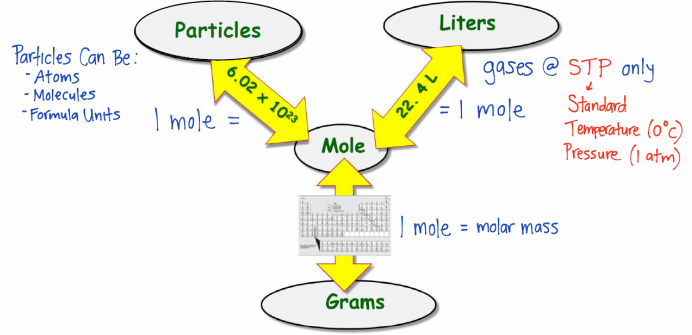
1 mole = 6.02 x 10^23 particles = molar mass (g) = 22.4 liters
Objectives
- Convert between the following units: moles, grams, particles, and liters
Before you can do molar conversions, you must know/be able to do the following:
- Calculations with scientific notation
- Round answers to the correct number of significant figures
|
References
Chapter 7, Section 3 Chapter 11, Section 3 (pg. 380)
|
Practice
pg. 85: Sample C Practice Problems #1-3 pg. 86: Sample D Practice Problems #1-3 pg. 87: Sample E Practice Problems #1-2 pg. 85-87_ANS KEY pg. 242: Sample I Practice Problems #1-3 pg. 381: Sample G Practice Problems #1-2
Problem #1 - Moles to Grams Problem #2 - Grams to Moles Problem #3 - Moles to Liters Problem #4 - Liters to Moles Problem #5 - Atoms to Moles Problem #6 - Moles to Molecules Problem #7 - Molecules to Grams Problem #8 - Grams to Liters
Mole WS #2 - Molar Mass #2 ANS KEY Mole WS #3 - Compound conversions #3 ANS KEY (scroll to page 3 for answers) |
PowerPoint & Videos
|
|
|
|