Molecule Shapes (VSEPR Theory)
|
References
|
Simulations
|
Tutorial Videos on Molecule Shapes
|
|
|
|
VSEPR - Valence Shell Electron Pair Repulsion
- Valence electrons (those in the outermost shell a.k.a. energy level) are involved in chemical bonding
- Valence electrons can be found in one of two types of electron regions:
- bonding region - electrons shared between two atoms
- lone pair region - group of 2 valence electrons that are not in a bond (a.k.a. not shared - attracted only to the center atom in the molecule)
- Electron regions repel one another due to similar charge
- Electron regions around the center atom in a molecule repel and move as far apart from one another as possible
- This REPULSION determines the shape of a molecule
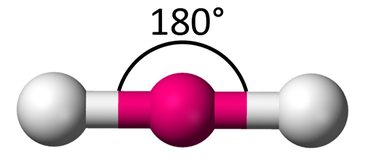
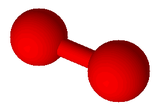
Linear
One Bonding Region
OR
Two Bonding Regions
One Bonding Region
OR
Two Bonding Regions
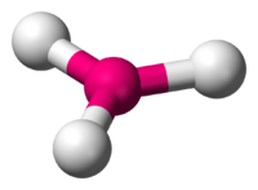
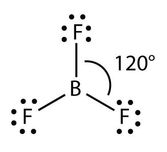
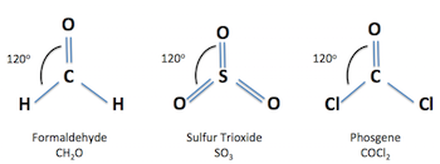
Trigonal Planar
Three Bonding Regions
Three Bonding Regions
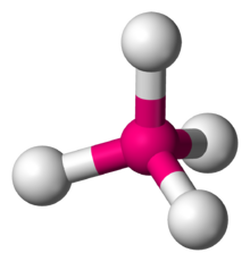
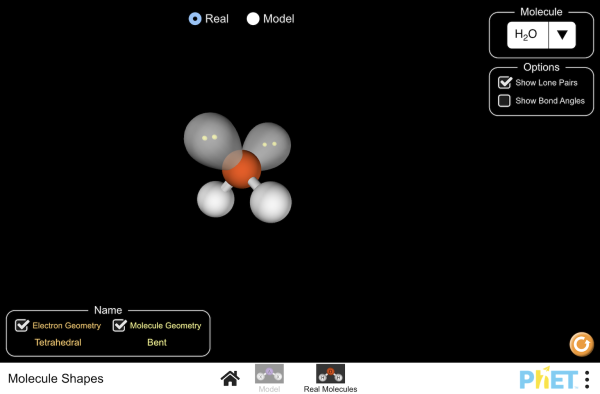
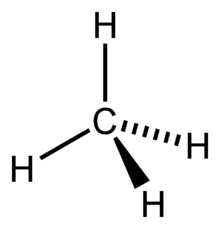
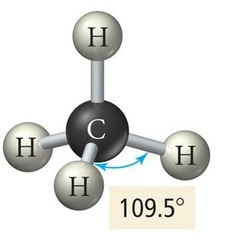
Tetrahedral
Four Bonding Regions
Four Bonding Regions
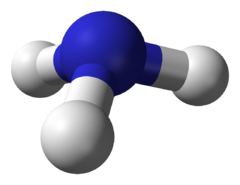
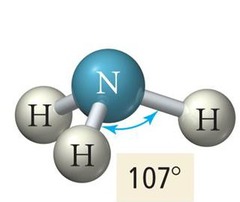
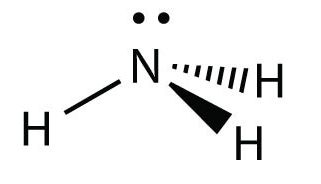
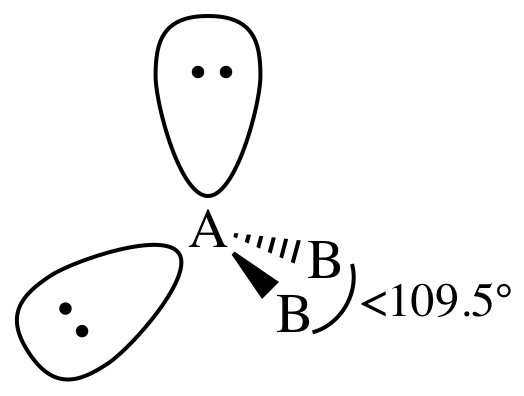
Trigonal Pyramidal
3 Bonding Regions + 1 Lone Pair
3 Bonding Regions + 1 Lone Pair
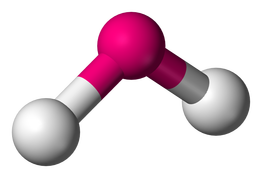
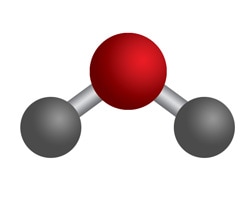
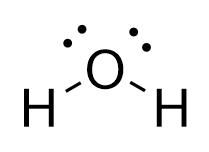
Bent
2 Bonding Regions + 1 Lone Pair
OR
2 Bonding Regions + 2 Lone Pairs
2 Bonding Regions + 1 Lone Pair
OR
2 Bonding Regions + 2 Lone Pairs